Improving retention rates in IBD clinical trials through remote testing
Improving retention rates in IBD clinical trials through remote testing
New treatments for inflammatory bowel diseases (IBDs) – such as Crohn’s disease and ulcerative colitis – must go through extensive and robust clinical trials before healthcare regulators will approve them as safe and viable alternatives to existing options. Accurate data is therefore vital for tracking the efficacy of a new drug in advanced therapy trials.
Faecal calprotectin is commonly used as a marker of inflammation and disease activity during IBD clinical trials, but lab-based testing requires patients to collect and store stool samples, then transport them to the hospital within a narrow timeframe. This is unpleasant, stressful and logistically challenging for patients, who may need to take time off work and spend money on transportation or parking, potentially while experiencing a disease flare. As a result, patients may postpone or avoid providing a sample, making it difficult to obtain linear trial data across multiple time points.
These issues are likely to contribute to the high participant drop-out rates from IBD clinical trials – estimated at around 20-30 per cent – with each lost patient reducing the statistical power of data, delaying trial completion and increasing overall costs. This illustrates the urgent need for an alternative, more convenient testing method that will encourage greater trial retention rates and support crucial research in the IBD space.
Could remote testing improve patient retention?
One solution could be to transition stool sample testing out of the lab to the point of care, enabling patients to test their samples themselves at home and bypass burdensome storage and transportation steps. Not only would this be far more comfortable for the individuals themselves, but it would also provide results far sooner than lab tests, streamlining data collection during trials. The COVID-19 pandemic normalised home testing with lateral flow tests, and digital health is now culturally embedded, paving the way for the adoption of this kind of decentralised testing across different disease areas.
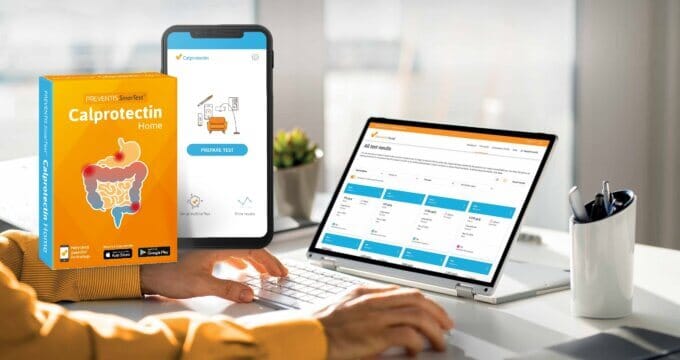
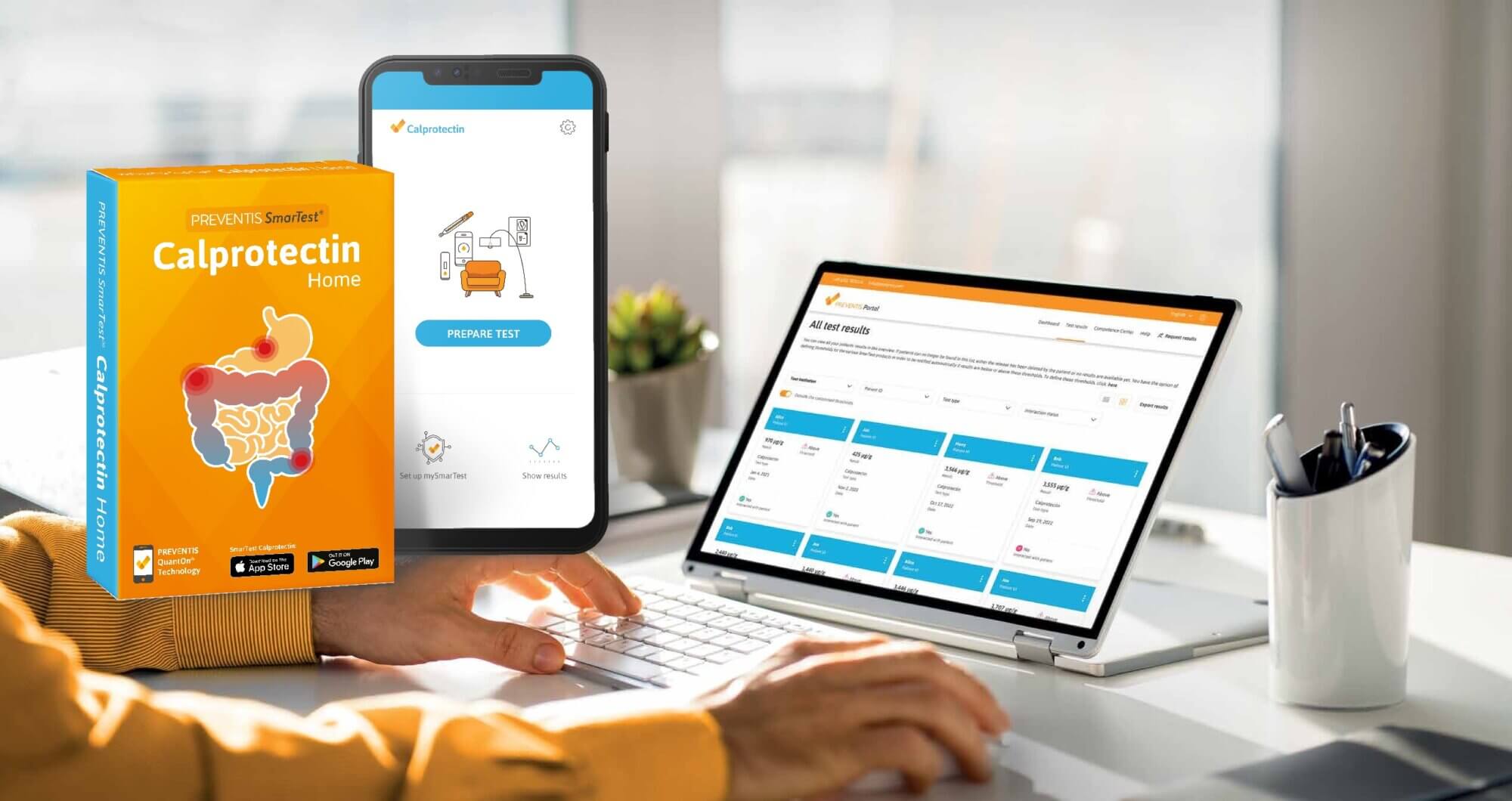
The SmarTest® Calprotectin Home test enables individuals to quantitatively monitor their disease activity at home with minimum test preparation or disruption to their day. The in vitro lateral flow test measures calprotectin using gold-conjugated anti-calprotectin antibodies, needing only very small quantities of sample that can immediately be flushed away. The assay is used in conjunction with a straightforward smartphone app, which rapidly and accurately quantifies the results, which are securely and automatically synced to an online portal, removing the need for manual data processing or data aggregation. This system allows patients and healthcare providers to evaluate drug effectiveness in real-time as part of routine monitoring or a clinical trial.
This type of testing is undoubtedly far easier and more hygienic for patients than collecting, storing and transporting stool samples on a regular basis. It minimises manual handling and provides clear, immediate feedback, which could help to improve the patient experience and, ultimately, increase the likelihood of trial compliance for improved retention rates.
The promising future of IBD clinical trials
Patient experience has a huge impact on the success of recruitment and retention in clinical trials. Testing at home without the need to transport samples removes many of the time and financial barriers associated with traditional lab-based faecal calprotectin tests, reducing the burden to IBD patients. This approach would also greatly streamline data collection, supporting faster and more effective trials. Remote testing with tools such as SmarTest therefore presents a fantastic opportunity to move data collection away from overwhelmed hospital labs and into the patient’s hands, encouraging long-term patient retention in much-needed clinical trials.
Learn more about the SmarTest Calprotectin Home test from BIOHIT HealthCare